
ViVitro Products at MDM West 2023, the largest medical device supplier show in the Americas. MDM West was still dealing with Covid restrictions in 2022. Last week’s show was the first to really connect with partners and grasp what is going on in the industry. It was super valuable.
One take away and hot topic was a slow down in the medical device industry. There is a markedly lower demand for equipment and services. This means it is even more important to work with an efficient company that will accelerate your development process.
In Our Booth
ViVitro Products at MDM West 2023 included the pulsatile flow capabilities of the SuperPump pulsatile pump along with a United Biologics TAVR Transcatheter Model with Replaceable Aortic Valves. They generated lots of questions specific for unique applications.

The ViVitro Labs Heart Valve Durability Tester with Automatic Dual Control (ADC™) technology attracted lots of interest for its modular, self-regulating approach.
Many visitors asked about strategies for device testing plans and how to identify critical path activities to minimize risks. You must have a proper plan before you start executing. That means adressing the highest risks early.
We were excited to discuss several opportunities related to customized flow loops with complex device indications.

Similary we had constructive conversations on the advantages of using an acute particulate matter evaluation with an in-line continuous particulate counting system. The importance of test method validation in the current regulatory environment.

But best of all, the show was a great opportunity to make new connections and reconnect with industry partners and medical device colleagues.
Please call (250-388-3531) or email us if you like to discuss your cardiovascular device engineering or testing needs. We look forward to talking with you in the near future.
Sincerely,
Karim Mouneimne
President – ViVitro Labs Inc
General Manager – ViVitro Labs SASU

ViVitro Products at MDM West 2023 highlight the ViVitro Heart Valve Durability Tester, ViVitro SuperPump pulsatile pump, Cardiovascular Medical Device Testing Services, and Custom Engineering for Cardiovascular Testing Equipment in booth 1570 on February 7-9, 2023.
 ViVitro Labs Heart Valve Durability Tester with ADC™ technology offers greater accuracy, less manual interaction saves time
ViVitro Labs Heart Valve Durability Tester with ADC™ technology offers greater accuracy, less manual interaction saves time
ADC automates individual test module stroke and pressure control.
- Dedicated linear electromagnetic motor utilizes position feedback for consistent
heart valve opening. - By-pass restrictor with patented control algorithm automatically maintains valve
differential pressure. - Continuous real-time data analysis counts every ISO cycle and yields 0% count error.
A prewired controller unit and data acquisition system accommodates any combination of up to 6 test modules.
Transparent customizable test chambers are designed for maximum flexibility to fit any valve size and geometry with optimum inflow, outflow, and lateral visibility on test samples.
ViVitro’s Heart Valve Durability Tester with ADC Technology has independent test modules that enable fast, easy interchangeability of test samples and system components with minimum disruptions to adjacent on-going test modules.
Automatic Dual Control technology maintains valve loading conditions throughout the entire test for up to 6 individually controlled chambers.
Flow Loop System autonomously runs under a low hydrostatic pressure head, which decreases the load on all seals and bellows. No external pressure source or compressor is required.
Contact us to learn more about the Heart Valve Durability Tester with ADC Technology and how ViVitro can help you meet your cardiovascular device testing needs.
ViVitro Heart Valve Durability Tester meets ISO 5840 AWT, DFM test requirements for all valve types and designs including: transcatheter and surgical valves:
aortic, mitral, pulmonary and tricuspid valve technologies, biological tissue valves, rigid metallic or polymeric and soft polymeric valve technologies.
ViVitro Heart Valve Durability Tester can be used to assess cardiac valve repair devices, transcatheter cardiac occluders, and cardiac implant devices.
ViVitro Heart Valve Durability Tester Features
- Automatic dual control system maintains optimum valve loading conditions during accelerated wear or real time testing.
- Optimum test chamber visibility for image capture in outflow, inflow, front, lateral, and 45° viewing positions.
- Large, customizable test chamber design fits all valve sizes and geometry and accommodates supporting structure(s).
- True independent test modules with no fluid connections and dedicated electronics including linear electromagnetic motors, automated by-pass control, and temperature measurement.
- Pressure design features low system mean (approx. 10 mmHg) for load reduction and increased lifespan of seals and bellows. No external pressure source or compressor required.
- Modular system optimizes part and component
- Standalone architecture eliminates neighboring module
- Vertical test sample configuration minimizes gravity effects on valve leaflets, leading to symmetric wear.
- Valve holder design facilitates chamber seal
- Integrated anti-vibration systems minimize pressure signal disruptions and improve high speed video quality.
- Inflow and outflow chamber annular compliance rings optimize differential pressure
- Independent heater and temperature sensors each test module and corresponding test valve.
- Adjustable intensity annular LED light delivers maximum high-speed imaging luminosity with minimal reflection impact.
- Customizable valve annulus ring fits all valve
- Adjustable outflow pressure sensor height accommodates different valve designs, including valves in conduits.
- Camera holder ensures repeatable high-speed camera positioning, increased test image capture quality and reliability between inspection
- Embedded safety systems include bellows leakage sensor, fluid level detector, temperature change and pressure drop monitoring.
- Sliding test module mechanism provides test sample inflow visualization and ease of servicing.

Software for ViVitro Heart Valve Durability Tester

The ViVitro Heart Valve Durability Tester with Automatic Dual Control Technology uses a proprietary software platform with a user- friendly interface. All in-software allows individual control of each test module.
Features
- Independent settings and controls for each test module, including stroke amplitude, fluid bypass control for differential pressure adjustment, test frequency, temperature, and many more
- Fully software-controlled system requires no manual adjustments
- Continuous and/or periodic data monitoring records ISO 5840 conformance as required for AWT and DFM.
- Easy control and settings navigation between test modules
- Integrated pressure calibration wizard
- Waveforms display for inflow outflow pressure, differential pressure
- Individual test module parameters display windows show passing cycles count, operating frequency, fluid temperature, peak to peak differential pressure, ¨% cycle pass, % pressure pass, and total number of passing cycles
- Test sample identification data embedded in test log with calibration values
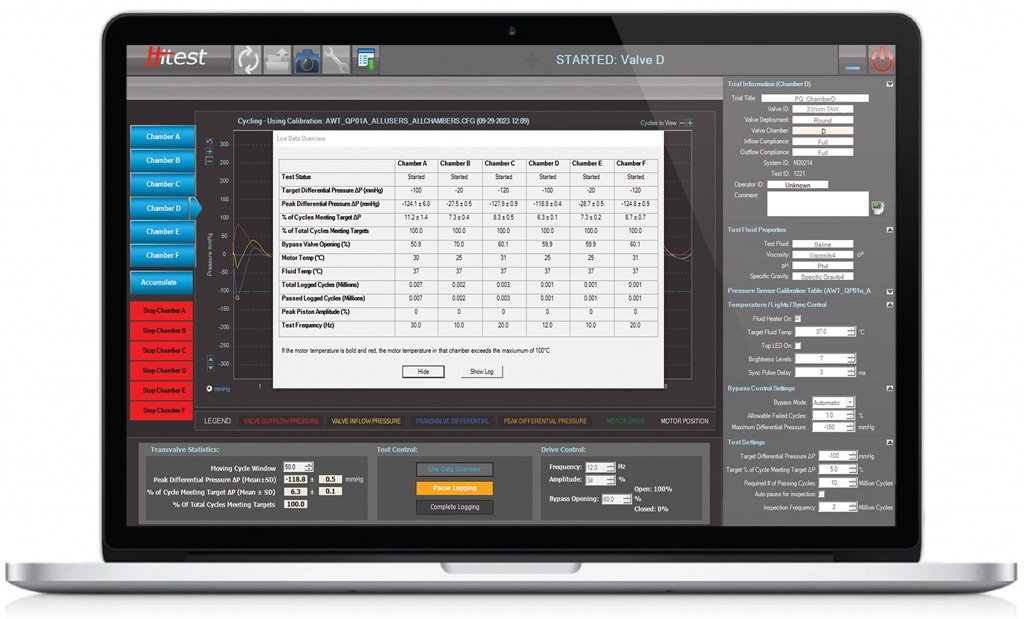
Continuous data log mode
Cycle to cycle information (peak differential, % of cycle meeting target pressure) can be collected for a predefined periods of time and save over multiple .CSV files. Data is recorded in engineering units.
Period data log mode
- Fixed number of cycles captured
- Periodic data report automatically generated based on user predefined parameters:
- Ability to periodically save entire waveform with user(s) defined output data (inflow and outflow waveforms minimum capture, and other test parameters)
- User-friendly field selector for output data
ViVitro Heart Valve Durability Tester Specs
Test module weight
Empty = 20.6 kg/module
Full = 22.1 kg/module
Full Controller and Data Acquisition unit weight = 8.4kg
Footprint – 6 module configuration
Min Bench Length = 1650 mm*
Min Bench Depth = 600 mm
Height = 362 mm (without Camera)
Height = 400 mm (with Camera)
Footprint – 2 module configuration
Min Bench Length = 850 mm*
Min Bench Depth = 600 mm
Height = 362 mm (without Camera)
Height = 400 mm (with Camera)
Controller power supply requirements fully loaded 675 W w/ 6 chambers with controller, 1 light ring, heaters on, at 100 mmHg No DAQ, No PC
System operating frequencies
1 Hz to 70 Hz
Test sample behavior and frequency response dependant.
Test chamber diameter
Max support OD: 95 mm
Test chamber height & clearance
Total Height/clearance up to 113.5 mm
Piston max stroke volume
38 ml/stroke
Test chamber transparency
Inflow, outflow, left and right, 1 angle at right 45°
Average hydrostatic pressure head
10 mmHg
*Minimal distance between units: 30mm
 中国地区询价
中国地区询价
 1 (250) 388-3531
1 (250) 388-3531
 +33 4 86 68 68 10
+33 4 86 68 68 10






