Experimental Flow Field Assessment – Digital Particle Image Velocimetry (DPIV)
The thrombogenic and haemolytic potential of the heart valve substitute can be assessed using a combination characterization techniques: digital particle image velocimetry (DPIV), computational fluid dynamics (CFD), and ex-vivo methods (e.g. blood loops).
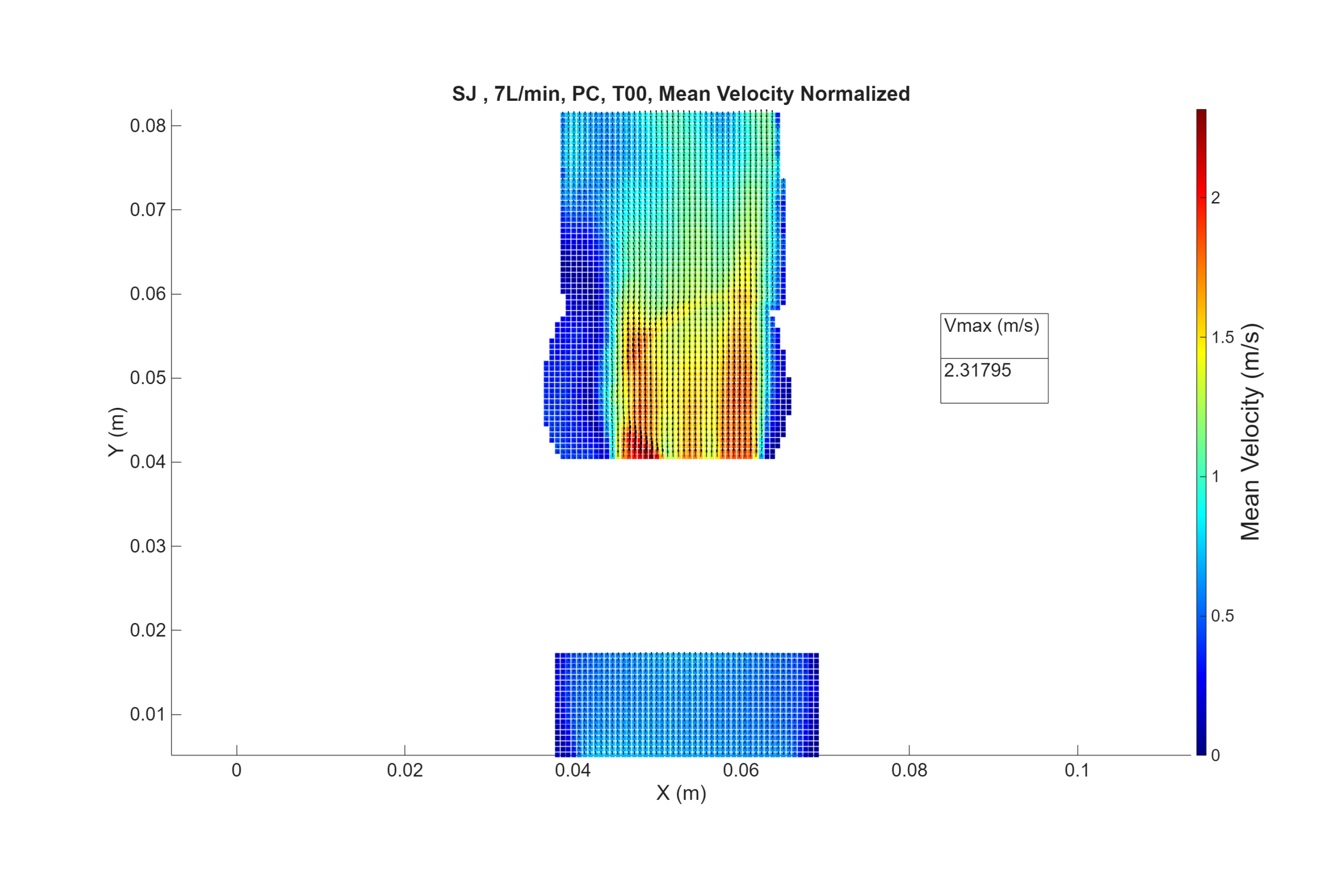
DPIV (using digital particle image velocimetry) is an experimental flow field assessment technique that it is used to determine areas of high shear stress and stagnation in the vicinity of prosthetic valve when subjected to hypo, normal an hypertensive flow physiological flow conditions. Under any flow conditions, when a device is positioned in a vessel, it will modify the natural flow field which can generate high shear stress (inducing haemolysis) and/or stagnation areas (inducing thrombosis).
The ViVitro Labs DPIV procedure and equipment can be used to test prosthetic valve and many other endovascular devices. DPIV test method has been validated with the ViVitro Labs Pulse duplicator using a standard configuration. Test method would need to be validated for other configuration.
Test Procedure
Velocity maps of the flow in 3 parallel planes in the immediate vicinity of the prosthetic cardiac valve with pressure and flow waveforms conditions consistent with a low and elevated cardiac output (e.g. 2 l/min and 7 l/min) at 70 beats per min as listed in table 3 of ISO 5840-1. A PIV Laser (65mJ, 15 Hz) is mounted on a traverse micrometric displacement system, and a camera is set perpendicular to the laser sheet. For each plane, image pairs (phase-locked, double-pulse images) are acquired every 0.0536 s (16 phases per cardiac cycle) and repeated 40 times (or more), which has been found sufficient to reach a statistical convergence for the velocities. The test fluid is seeded with particles. Particle area density is controlled, and pulse separation time (∆t) is adjusted to ensure quality cross-correlation results. The PIV system is synchronized with the pressure-flow measurements using an external trigger delivered by ViVitro SuperPump controller.
Test Method Validation
- An interoperator study was performed with 2 operators and 2 PIV calibrations. Each operator captured 10 groups of 40 cycles at nominal conditions.
- Velocities measured by the PIV system were converted to flow and compared to the results of the flowmeter. This analysis was performed on the interoperator data that was used for the measurement uncertainty.
- Factors influencing the results were identified and controlled: Temperature of test fluid, Particle density, Particle displacement and S/N ratio, Fluid viscosity, Time delay between images, Laser and camera alignment, Flow rate, Ambient light.
- Using the results of the interoperator study, an uncertainty budget was defined for the measurement of velocity by the PIV system. The expanded velocity uncertainty with coverage factor k=95% is ±0.07m/s
Equipment
- ViVitro Pulse Duplicator + specific valve fixtures (specific site section)
- Aortic PIV test chamber or other defined by study sponsor
- Aortic PIV test chamber (surgical valve only) – customization required for any other device
- Laser, 65 mJ, 15 Hz, Litron, Nano S 65-15 PIV
- Camera, Andor Zyla 5.5
- Light sheet optics 1.5” BSL/NWL/Evergreen
- PIV software DynamicStudio (current version)
- Synchronizer
- Computer
Measured Parameters
- Velocity
- Shear stress (viscous shear stress, Reynolds shear stress)
- Regions of stagnation
- Qualitative analysis: Turbulent flow region, flow separation, vortex formation, jets
- Calculated particle residence time (if required)
Scope
Applications
- Transcatheter valves; TAVI, TMVR
- Biological, Polymeric, Mechanical valves: Rigid or soft
- Venous valves and conduits
- Valvular conduits
- Vena Cava Filters
- Assist ventricular devices
- Any device implant in flow models
Service Levels
Applicable Standards
-
ISO 5840-2:2021
Cardiovascular implants — Cardiac valve prostheses — Part 2: Surgically implanted heart valve substitutes -
ISO 5840-3:2021
Cardiovascular implants — Cardiac valve prostheses — Part 3: Heart valve substitutes implanted by transcatheter techniques -
ISO 5840-1:2021
Cardiovascular implants — Cardiac valve prostheses — Part 1: General requirements
Related Tests
- Pulsatile Flow Testing – Hydrodynamic Performance
- Steady Flow Testing – Hydrodynamic Performance
- Accelerated Wear Testing (AWT)
- Experimental Flow Field Assessment – Echo Doppler – Bernoulli’s Equation Verification
OLE0914

Simplified PIV setup including laser, lenses, high speed camera, and PIV Load Assembly in a Pulse Duplicator.


ViVitro Labs Advantages
Many companies look to academia to handle their testing needs due to the reduced cost. With ViVitro Laboratory Services you gain the following advantages:
ISO 17025 Accredited
For studies used for regulatory approval this is a must.
Victoria and
Marseille labs accredited by A2LA.
Test Method Validation
The FDA and Notified Bodies around the world are requiring evidence of Test Method Validation.
ViVitro Labs has invested significant resources to validate the most common test methods that are not ISO 17025 accredited.
We continue to work closely with device manufacturers and share the burden with our clients to validate other specific test methods that are less in demand.
Timeliness
We understand how critical your timelines are.
Our expertise and experience ensure your schedule is met and our commitments are delivered on time.
Free Initial Consultations
All of our studies are scoped via no-cost, no obligation, collaborative consultation.
Guaranteed Confidentiality
We happily provide Non-Disclosure Agreements for all services, including initial consultation.
 中国地区询价
中国地区询价
 1 (250) 388-3531
1 (250) 388-3531
 +33 4 86 68 68 10
+33 4 86 68 68 10
