ViVitro Labs
Our team brings extensive cardiovascular device testing experience to accelerate the device development process while increasing patient safety with enhanced conformity to medical device standards.
Our engineered solutions are designed to meet a wide variety of cardiovascular test applications and requirements. Our scalable and modular test systems can be adapted to cover evolving program needs from fundamental research to early-stage to device Validation and Verification (V&V activities) while producing highly reliable results. Our test methods are fully transferable with turnkey pre-configured or customized test solutions combining validated test methods with modular and versatile test systems.
We offer:
- Scalable and modular test systems to assess the performance of devices under real, physiological, and accelerated conditions.
- Reliable, repeatable, reproducible and scalable (RRRS) test methods to de-risk the device development process from proof of concept to regulatory submissions.
- Scalable validated test methods that can be integrated into any organization at any time during the device development process via transfer of knowledge of our proprietary test procedures and protocols.
How we do it
We empower engineers and medical device manufacturers to become self-sufficient and accelerate knowledge transfer through validated test methods and advanced training programs. By combining high-quality test systems with ISO 17025–validated test methods, our turnkey engineering solutions enable the rapid deployment of cost-efficient testing programs that deliver highly reliable results.
We leverage our ISO 17025–accredited laboratory to enhance the reliability of medical device performance testing by transferring repeatable, reproducible, and replicable validated test methods in combination with our test systems. Our test systems are manufactured in accordance with ISO 13485 processes to optimize equipment quality and reliability.
Drawing on our ISO 17025 laboratory expertise, we develop forward-looking test methods and test systems tailored to cardiovascular research and medical device innovation needs. Our extensive cardiovascular testing experience helps customers navigate the complexity of in-vitro testing programs by defining test strategies that optimize success while de-risking the device development process.
As an active member of several ISO and ASTM working groups, ViVitro Labs remains at the forefront of innovation and regulatory evolution..
ViVitro Labs management and employees bring a wealth of experience and technical expertise in cardiovascular device testing, test equipment and test method development. We provide our clients sustainable and reliable engineering solutions that further research projects and advance medical device development.

Karim Mouneimné
Karim Mouneimné brings over 25 years of medical device industry experience in Europe and the US. He has held commercial development and R&D positions with manufacturers including Johnson & Johnson and Edwards Lifesciences. Karim founded Venture Medical Solutions (VMED) and served as Managing Director until the company was acquired by PROTOMED in 2008. The architect, President and CEO of ProtomedLabs, an ISO 17025 accredited laboratory specialized in cardiovascular device performance testing, Karim adds a global perspective with deep technical and client knowledge to ViVitro leadership. Karim holds a Bachelor and Master of Science in Biomedical Engineering from Arizona State University.

Vincent Garitey
Vincent Garitey has over 25 years of experience in the medical device industry. He holds several device patents with French Key Opinion Leaders. As a former AFNOR and AFSSAPS expert, Vincent reinforces ViVitro Labs leadership position in cardiovascular device testing. Vincent played a major role in establishing ISO standard specifications for cardiac valve prosthesis. He spent 5 years as a scientist for MecaBio specializing cardio-vascular devices research and was the lead engineering in the development of Prof. Alain Carpentier’s mechanical valve prosthesis, a collaborative project with BAXTER Healthcare and SICN. Garitey holds a PhD in Fluid Mechanics from Université de la Méditerranée.

Troy Nickel
Troy brings extensive experience in cardiovascular device testing including director positions in R&D, new business development and product management. Prior to joining ViVitro, Troy was Senior Product Manager at TA Instruments where he led product strategy & marketing and application support for ElectroForce mechanical test systems. He has led design projects, design teams, technology research, and voice of customer projects at EnduraTEC Systems, Bose Corporation/ElectroForce, and TA Instruments. Troy holds a MS in Mechanical Engineering from the University of Minnesota.

Janet Murray
Janet Murray brings 14 years of experience in Operations Management, Sales, and Customer Service for diverse industries including software and publishing. As Operations Manager, she leads all aspects of Client Relations and Technical Support, managing order processing and customer post-order communication. Janet interfaces with various cross-functional groups and departments to ensure quality and on time delivery of ViVitro offerings. She is committed to growing service offerings and ensuring a high level of customer satisfaction. Janet holds a Bachelor of Arts with a focus in English from Concordia University.
A history of innovation, a reputation for excellence.
ViVitro Systems was founded by David Walker, Ph.D. and Larry Scotten, Dipl. T. in the mid-1970s. Their initial challenge was to build test equipment to evaluate a heart valve developed in the Cardiac Development Lab at Victoria’s Royal Jubilee Hospital. David and Larry’s prototype (built using a VW piston) was soon able to simulate valve positioning and physiological compliance accurately and repeatedly. Further development resulted in the first commercial Pulse Duplicator in 1984. The team later developed the HiCycle accelerated durability tester – a system that became recognized as the world’s most efficient and cost effective means of assessing heart valve durability.
In 2009, ViVitro Systems assets were acquired by StarFish Group and renamed ViVitro Labs. StarFish Group is a Victoria BC-based company that includes StarFish Medical, a medical device design development and manufacturing firm. Their co-located relationship enables ViVitro to leverage extensive medical device development expertise and quality systems.
Current Team

2025
ViVitro acquires ProtomedLabs SASU

2019
Endovascular Simulator

2016
Larry and David view the ViVitro museum with Gerry Wight, GM.

2015
Fortis Hitech Ltd is signed as China distributor.

2014
ViVitro Labs testing services earn ISO 17025 accreditation for Durability Testing, Pulsatile-Flow Testing, and Steady Flow Testing for Haemolytic and Thrombogenic properties of Device Design.
2012
Pulse Duplicator, SuperPump and HiCycle Advanced Research (AR) Series introduced migrating system from analog to digital. Lower voltage controller improves safety.
2011
ViViTest upgrade: I/O module is created replacing the VIVIBOX connector Box PC.

2010
ViVitro products and assets are acquired by StarFish Medical.

2009
StarFish Medical begins manufacturing some products for ViVitro Systems.

2007
ViVitro Systems Inc at Royal Jubilee Hospital.

1998
First presentation describing the SuperPump at AAMI meeting in Dallas, TX with Larry Scotten.
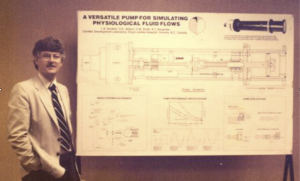
1983
First SuperPump

1982
Bubble-shaped leaflets
made from bovine pericardium.
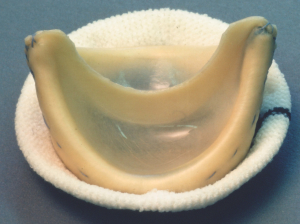

1981
 中国地区询价
中国地区询价
 1 (250) 388-3531
1 (250) 388-3531
 +33 4 86 68 68 10
+33 4 86 68 68 10







